 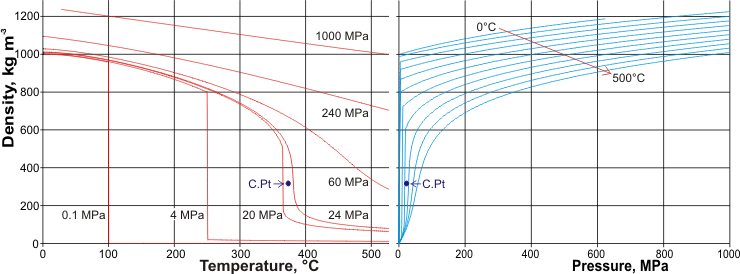
Sometimes, you have to convert units to get the correct units for density, such as mg to g or in3 to cm3. For example, if you had a metal cube with mass 7.0 g and volume 5.0 cm3, the density would be. There are exceptions however, such as water's density increasing between 0☌ and 4☌.īelow is a table of units in which density is commonly expressed, as well as the densities of some common materials. To solve for density, simply follow the equation d m/v. Density is a measure of the amount of mass contained in a unit of volume.The general trend is that most gases are less dense than liquids, which are in turn less dense than solids, but there are numerous exceptions. one gram per milliliter ( cm3) at STP The mass unit of the metric. This definition connects the basic units of mass, volume and distance in the metric system ( brilliant) As 1 gram of water occupies one ml ( 1cm3), the density of water is 1 g ml. Increases in temperature tend to decrease density since the volume will generally increase. Heres a table of densities of common substances, including several gases, liquids, and solids. Explanation: The mass unit of the metric system was defined as the mass of one cm3. An increase in pressure decreases volume, and always increases density. However, when regarding gases, density is largely affected by temperature and pressure. At a temperature of 15 degrees Celsius and a salinity of 35 parts per thousand (ppt), the density of seawater is approximately 1023 kg/m³. However, this can vary depending on the salinity and temperature of the water. In the case of solids and liquids, the change in density is typically low. The density of seawater typically ranges from 1020 to 1030 kg/m³. The density of a substance is the same regardless of the size.  /beaker-containing-layers-of-fluid-of-different-densities-including-water-and-oil-and-objects-float-93604846-57a7687c3df78cf45916180b.jpg)
The density of water is 1 gram per cubic centimeter. Just like a solid, the density of a liquid equals the mass of the liquid divided by its volume D m/v. Note that density is also affected by pressure and temperature. The mass and size of the molecules in a liquid and how closely they are packed together determine the density of the liquid. It is useful to carefully write out whatever values are being worked with, including units, and perform dimensional analysis to ensure that the final result has units of mass There are many different ways to express density, and not using or converting into the proper units will result in an incorrect value. The same density of water is 1.0249 g/ml or 63.982 lb/ft. And that's it The density of salt water is 1,024.9 kg/m. However, it is important to pay special attention to the units used for density calculations. What is the density of water at 20 C of salinity S 35 and under the pressure of 1 atm Let's give our water density calculator a try and find out Set the temperature to 20 C. The density of water is 1 g per cm3 and density of water in kg per m 3 is 977 kg per m 3. Acrolonitrile, 25, 801 Alcohol, ethyl (ethanol), 25, 785.1 Alcohol, methyl (methanol). A substances density is defined as its mass per unit volume. Densities of common liquids like acetone, beer, oil, water and more. Also, pure water is less dense than seawater, so fresh water can float on top of salt water, mixing at the interface. A consequence is that ice floats on water. Unlike most substances, water is denser as a liquid than as a solid. It is odourless, tasteless, clear, and colourless. Note that the density of pure water is defined to be 1 gram per cubic centimeter (or, g/ml). In order to convert the cm 3 in the denominator of 0.The calculation of density is quite straightforward. Water is the principal ingredient of this planets lakes, oceans, streams, and the fluids of most living species. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
